The Paclitaxel eluting balloon luminor 35 is a double lumen catheter from the connector to the tip (also called over-the-wire, OTW) designed for percutaneous transluminal angioplasties of large peripheral arteries. The guidewire runs into the circular-largest lumen while the elliptic-smallest lumen is intended for contrast medium flow. The maximum diameter of the guidewire must not exceed 0.89 mm = 0.035 inches. On the catheter’s distal part, just before the tip, there is the balloon (inflatable segment) that will dilate the artery upon inflating by means of infusion of contrast fluid inside it. The connector is Y-shaped and it has two entry ports: The straight port is intended for guidewire passage. The side port allows contrast medium flow to dilate the balloon The balloon is coated with a homogeneous mixture of Paclitaxel, a derivative of Taxol, and a physiologically innocuous matrix, the excipient. The drug’s dose is 3 µg/mm2 of balloon surface and it is intended to avoid cellular proliferation, consequently decreasing the re-intervention rate. The drug is released from the balloon by means of rapid inflation so that a high dose is released in a very short period of time. In order to assure a sufficient dosage of Paclitaxel onto the arterial wall, the inflation process must last from 30 seconds to 1 minute. Dilation of the lesion can be optimized by using longer inflation times at the operator’s discretion. The balloon is designed to reach different diameters at different pressures, as predicted by the compliance curve included on the primary packaging. Two radiopaque markers of Pt/Ir alloy are located at each end of the balloon in order to mark its length and help the user to see the catheter while navigating inside the patient. At the distal end, there is the tip, made from a very soft polymer and rounded in a non-traumatic shape in order to avoid damaging the arterial wall while navigating. The shaft of the catheter is coated with a proprietary hydrophilic formula in order to minimize friction See IFU for further information. Available to download.
Main features:
- Over the wire catheter (OTW)
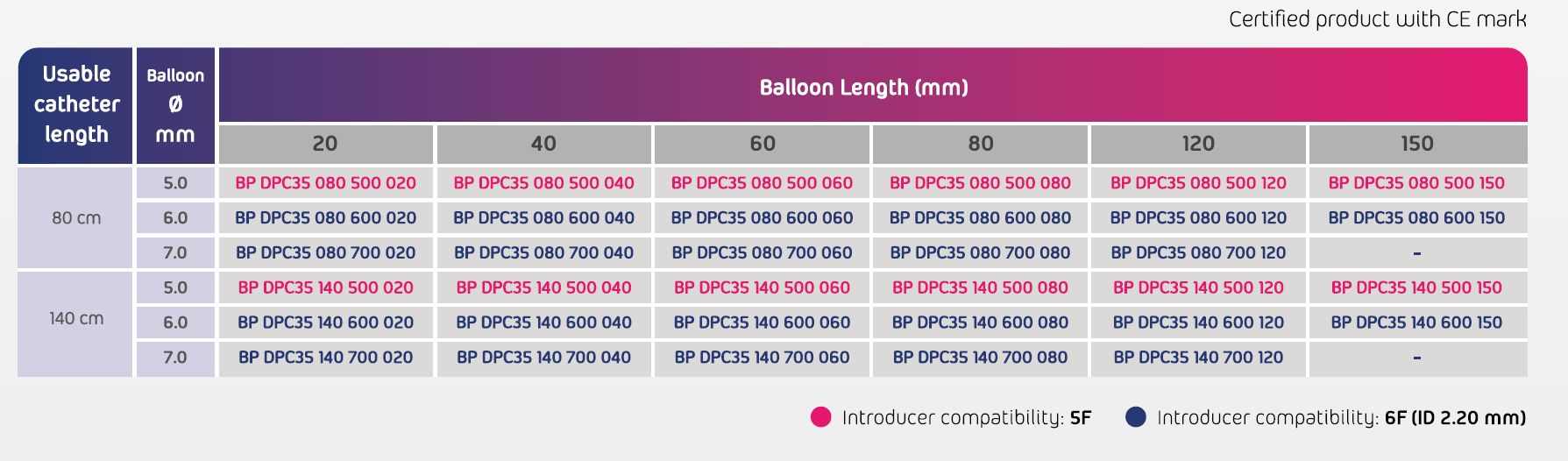
- Usable catheters length: 80 & 140 cm
- Compatible with 0.035" guidewire
- Crossing profile: from 0.065" up to 0.077"
- Tip profile: 0.036"
- 2 radiopaque Markers
- Np: 6/7atm | RBP: 16 atm | ABP: 23 atm