The peripheral stent “Restorer” is made of a Cobalt-Chromium alloy called L605. The stent is supplied pre-mounted on a delivery system for its implantation at the lesion to treat using the balloon's expansion at its distal end. The device is designed for percutaneous transluminal angioplasties of peripheral arteries. The stent is designed to be implanted in different artery diameters by adapting its open-cell design with alternating connection points. The stent is manufactured by laser cutting of a metallic tube, and then it is submitted to several treatments to reach a smooth and glossy surface finish. The stent design is based on circumferential cells linking, which are axially interconnected using bridging links that provide different longitudinal configurations. The stent delivery system is a balloon catheter with a double-lumen configuration and OTW architecture. It features a double-lumen tube from the proximal connector to the balloon. One lumen is for guidewire insertion, which will lead the catheter to the lesion. The other one comprises the inflation channel, which allows contrast liquid to flow to inflate the balloon. The guidewire diameter must not exceed 0.89 mm = 0.035’’. At the proximal end is located the connector with two entry ports, one for a balloon in/deflation and the other for guidewire passage. The catheter shaft surface is homogeneously coated with a lubricious silicone-based coating to minimize friction and facilitate navigation through vessels. See IFU for further information. Available to download.
Main features:
- Over the wire catheter (OTW)
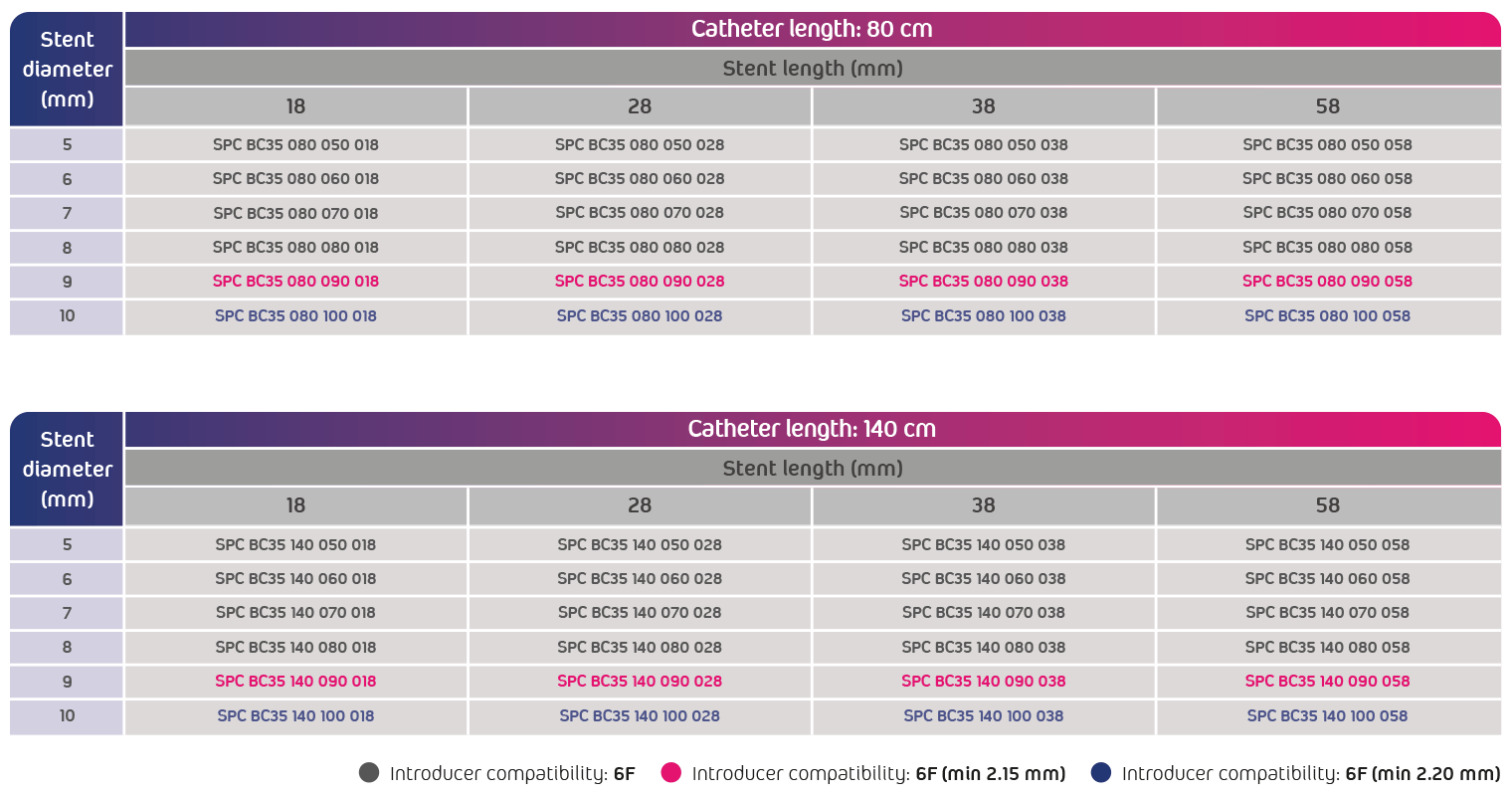
- Compatible with 0.035" guidewire
- Compatible with 6F introducer
- Tip profile: 0.036"
- 2 radiopaque Markers
- Np: 10-12 atm | RBP: 16 atm (Ø 5-6-7 mm stents), 15 atm (Ø 8 mm stents), 14 atm (Ø 9-10 mm stents)
- Stent material: CoCr L605
- Recoil <6%
- Shortening at expansion < 3%